 A sustainable energy supply is a major challenge for humanity in the 21st century. Innovative materials are needed to not only convert renewable resources into useable energy forms, but to also store that energy so it can be reliably accessed at any time. Professor Lianzhou Wang is Director of the Nanomaterials Centre at AIBN, where his research focuses on the design and development of functional nanomaterials to achieve these aims.
A sustainable energy supply is a major challenge for humanity in the 21st century. Innovative materials are needed to not only convert renewable resources into useable energy forms, but to also store that energy so it can be reliably accessed at any time. Professor Lianzhou Wang is Director of the Nanomaterials Centre at AIBN, where his research focuses on the design and development of functional nanomaterials to achieve these aims.
Understanding the relationship between the physical structure of a battery’s materials and its electrochemical performance is a major aspect of Professor Wang’s research and he believes it is crucial to future advances in energy storage technologies.
Recently he teamed up with researchers in academia and industry to develop printed flexible batteries. A key advantage of such technology that it can be adapted to take almost any shape, yielding a wide range of potential applications from wearable electronics to large scale energy storage. Of course, such a versatile physical structure presents new challenges and opportunities with regard to chemistry and design.
 Professor Wang, together with Dr Dai-Wei Wang of UNSW and Professor Chris Greig of the UQ Dow Centre for Sustainable Engineering Innovation, will work with Printed Energy, Sunset Power International, and Sonovia Holdings to research and develop the new technology. This collaboration has just been awarded $2 million in CRC-Project funding by the Australian Government through the Collaborative Research Centres Programme.
Professor Wang, together with Dr Dai-Wei Wang of UNSW and Professor Chris Greig of the UQ Dow Centre for Sustainable Engineering Innovation, will work with Printed Energy, Sunset Power International, and Sonovia Holdings to research and develop the new technology. This collaboration has just been awarded $2 million in CRC-Project funding by the Australian Government through the Collaborative Research Centres Programme.
“We are very pleased to have this opportunity to work with the leading Printing Technology industry partners on this exciting program, addressing some critical energy storage challenges,” says Professor Wang.
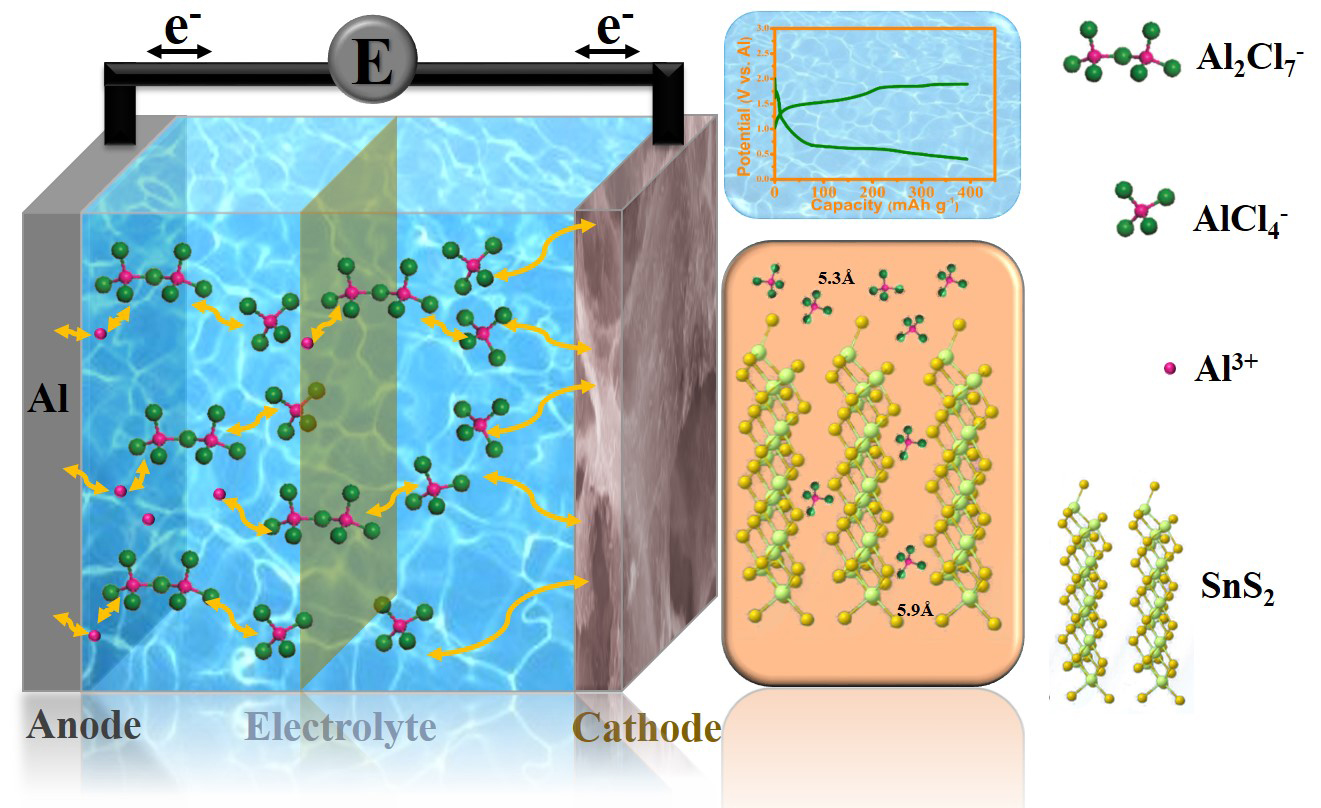
By advancing our knowledge of the structure-function relationship in batteries, Professor Wang’s group is also improving the design of rechargeable aluminium ion batteries. This could have an important impact on the energy storage sector, and by extension, influence the shift toward renewable energy sources.
While lithium is commonly used in rechargeable batteries, aluminium is safer, more abundant, and more affordable. Professor Wang and his colleagues Mr Yuxiang Hu and Dr Bin Luo believe aluminium batteries also have the potential to compete with lithium batteries in terms of energy and capacity.
Limited progress has been made with aluminium batteries thus far mainly due to the formation of a passive oxide film and intrinsic hydrogenation over the aluminium metal.
“This creates a barrier between the electrolyte and the cathode, which leads to serious reduction of the cell voltage, capacity and efficiency,” says Mr Hu.
In any battery, the chemical reactions that take place on the electrodes create the flow of electricity, and the electrolyte’s job is to shuttle ions between the two electrodes while this is going on. Consequently, it’s important these components work well together.
Recently, researchers in the US used a graphite foam as the cathode, and also used a novel aluminium liquid which did not form a problematic barrier between the electrode and electrolyte. The resulting aluminium batteries were able to charge quickly and could be recharged many times. Unfortunately, when it came to storage capacity they didn’t perform very well.
To find out why, AIBN’s Professor Wang and his team took a closer look at the structure of the graphite and determined that the space between layers of graphite were simply too narrow. This meant that the relatively large chloroaluminate anions of the electrolyte had trouble getting between the graphite layers. The tight fit slowed down the process of shuttling ions between electrodes, reducing battery storage performance.
“The structure of the materials decide the performance of the battery,” says Professor Wang.
Inspired, he and his team designed a new cathode structure using graphene instead of graphite. Graphene is a sheet of repeating carbon molecules, and under the right conditions the researchers were able to create a three dimensional matrix of criss-crossing graphene sheets. The key advantage of the graphene was that it served as a conductive support structure for numerous tin sulphide (SnS2) nanosheets.
Chloroaluminate anions can easily exchange charge with tin sulphide and the multi-layered nanosheets of tin sulphide provide plenty of opportunity for interaction. Their unique structure also provides more room for the bulky chloroaluminate ions to move around.
Tests show that the 3D graphene-supported tin sulphide material works extremely well as a cathode. It has high storage capacity, and also exhibits very good electronic conductivity with high charging speed. The Wang group’s research has been published in the journal Advanced Materials.
“This provides a new understanding of the charge-discharge mechanism, which is an important consideration in the design of aluminium ion batteries,” says Professor Wang.
So not only is the new electrode material useful in its own right, this research will also help inform future advances in aluminium battery technologies for a more sustainable future.



