Researchers at UQ’s Australian Institute for Bioengineering and Nanotechnology have discovered that hard nanoparticles can enter human cells, including cancer cells, much more easily than soft ‘squishy’ nanoparticles. The findings will help to guide the design of more potent, highly targeted treatments for a range of diseases.
Targeted drug delivery is a major challenge in the development of new treatments. Scientists have long sought a way to deliver medicine to cells that need it while avoiding cells that don’t.
Fortunately, recent advancements in nanotechnology are proving incredibly useful in this area. For example, nanoparticles can have the potential to deliver a cargo of medicine to where it’s needed in the body, with the help of specialised molecules that act a bit like a postal address.
In the case of cancer treatment, nanoparticles can deliver a concentrated dose of chemotherapy treatment directly to tumour cells, while leaving healthy cells alone. In the case of autoimmune diseases like rheumatoid arthritis, nanoparticles may be able to deliver anti-inflammatory molecules directly to the specific immune cells that have run out of control.

Once the nanoparticle arrives at its destination, it needs to enter the cell as efficiently as possible, explains AIBN researcher Associate Professor Chun-Xia Zhao. In other words, we don’t want the package to just reach the front door, it also needs to get into the house.
"It is known that the physical properties of nanoparticles, such as size, can influence this entry process," says Assoc Prof Zhao.
She suspected that stiffness may also play a role. After all, it’s known that the stiffness of a virus particle can change over its life cycle, and this seems to influence how well the virus can enter or exit a cell.
“Mature HIV particles become softer to facilitate their entry into host cells,” Assoc Prof Zhao explains, “While newly formed, immature HIV viruses are more rigid for budding out of a host cell.”
“It is of great importance to understand how cells sense and respond to such external mechanical cues at nanoscale,” says Zhao, explaining that such knowledge would be a big advantage when designing nanoparticles. “Unfortunately, this is largely unknown.”
So she and her colleagues decided to find out if the stiffness or ‘squishyness’ of a nanoparticle affects how well it enters cells. And it turns out, it does, but not quite in the way they expected.
The main challenge was to find a way to change the stiffness of a nanoparticle while leaving every other variable, such as size, alone.
“We developed a patented bioinspired nanotechnology for producing nanocapsules, using designed peptides under room temperature, neutral pH and without requirement of any solvents or toxic chemicals,” says Zhao.
This enabled the team to create the first ever library of nanocapsules with a wide range of stiffness, ranging from highly elastic to highly rigid.
They then examined how well each particle was able to enter different cell types, including cancer cells and immune cells called macrophages.
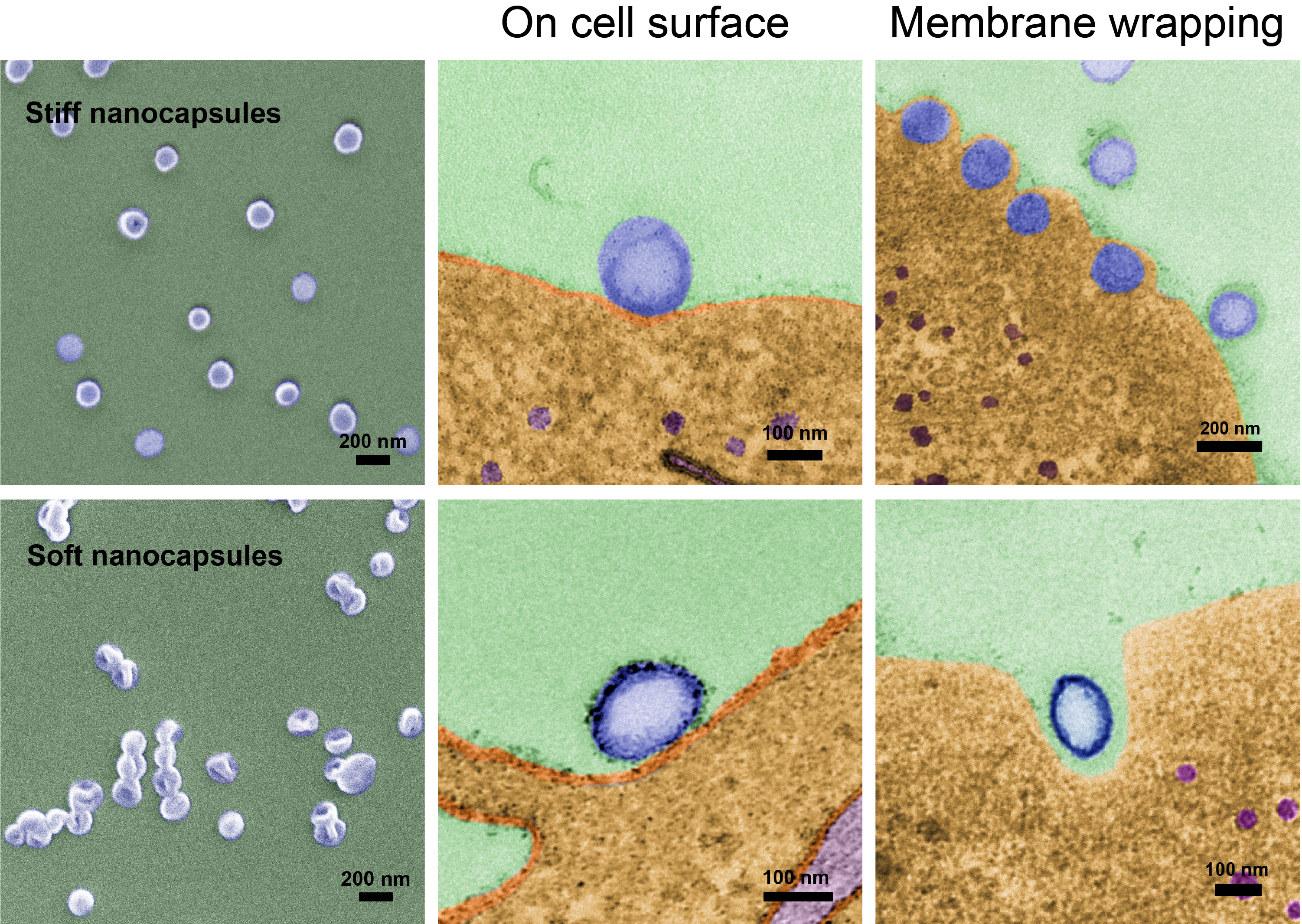
Whereas mature HIV virus particles are softer and more flexible when entering a cell, Zhao and her team discovered that ‘squishy’ nanocapsules did not enter cells as well as stiffer nanocapsules.
High powered electron microscopy revealed why. It turns out that the stiff nanocapsules could remain spherical during cellular uptake, whereas the soft nanocapsules became deformed by forces encountered when docking with a cell and being enveloped by the cell membrane.
“This deformation in turn reduced the cellular binding and the entry rate of the soft nanocapsules,” says Zhao.
“This work demonstrates the crucial role of the elasticity of nanoparticles in modulating their entry to macrophages and cancer cells, and may shed light on the design of drug delivery vectors with higher efficiency.”
The findings have been published in Science Advances.


