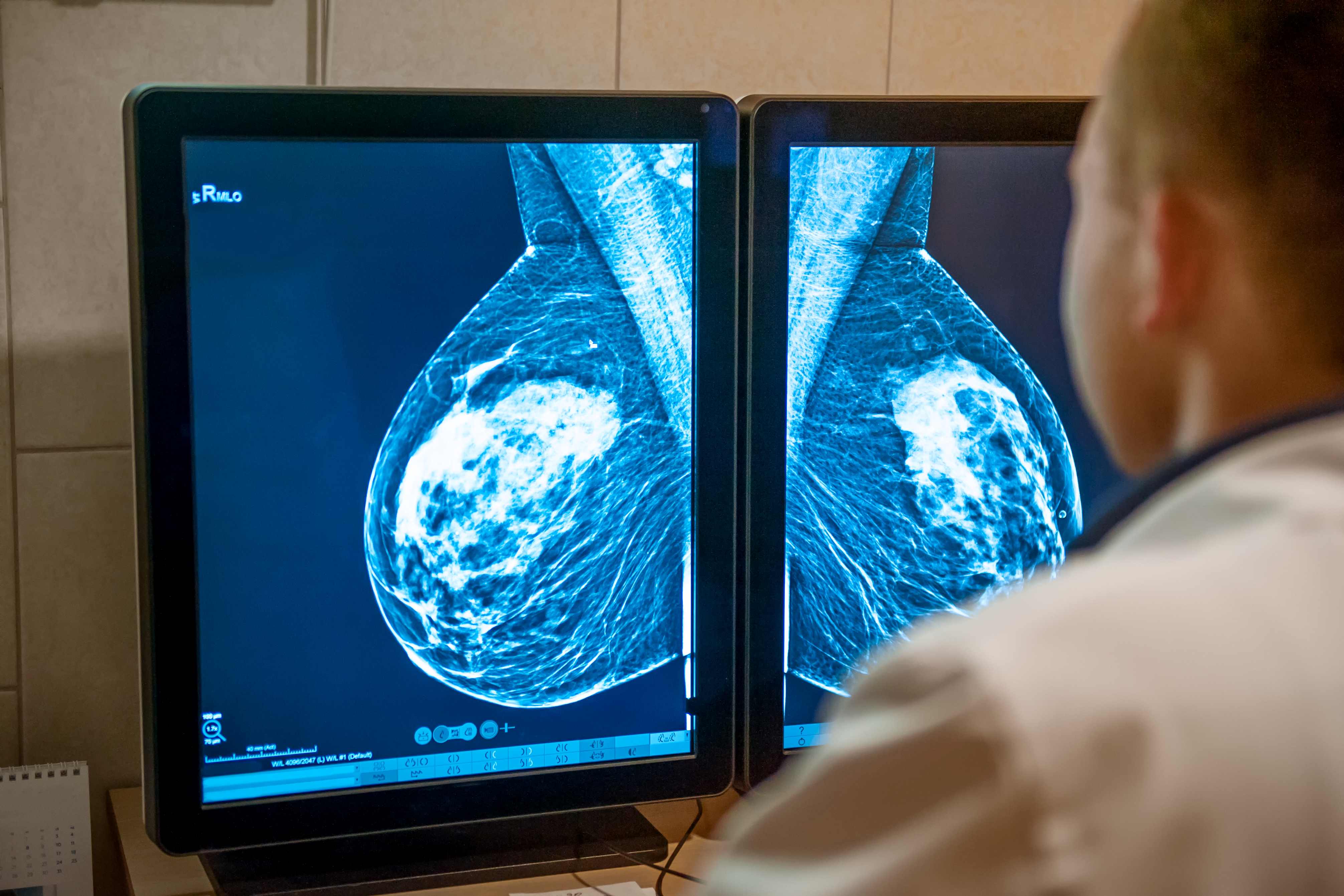
Researchers at the Australian Institute for Bioengineering and Nanotechnology (AIBN) and The School of Chemical Engineering believe decoding a cellular messaging system will give them the upper hand against a particularly deadly type of breast cancer.

Triple-negative breast cancer is aggressive, harder to treat and - for the 2500 Australian women who are diagnosed each year – the disease is more likely to spread to other organs and result in death.
Associate Professor Joy Wolfram says this is partially because triple negative breast cancer cells are able to hijack a messaging network in our bodies and convince the immune system to ignore the problem. Stay on top of our industry news and developments, events and opportunities, by joining The NetworkJoin The Network
Now, with the help of a $590,452 National Breast Cancer Foundation grant, Associate Professor Wolfram hopes to find out exactly how this hijack takes place, and what we can do to intervene.
“We know that our immune system plays a critical role in detecting and eliminating breast cancer cells,” Wolfram says.
“But triple negative breast cancer cells have a trick. They release biological text messages that tell the immune system to look the other way, allowing the cancer to grow and spread more aggressively.
“We aim to find out how this happens.”
Associate Professor Wolfram said it is important to note that these internal text messages - known as extracellular vesicles (EVs) - are not necessarily bad on their own.
In fact, researchers are increasingly confident EVs could be key to the development of new treatments and therapies because of their ability to facilitate communication between cells.
Associate Professor Wolfram says the problem was EVs secreted by aggressive cancer cells.
“EVs have potential to provide a paradigm shift in cell-free therapy, but we know they are a double-edged sword,” she says.
“When they are secreted by triple negative breast cancer cells, they have the ability to promote immunoevasion, allowing the cancer cells to survive and spread to other organs in the body.”
With assistance from co-investigators at the UQ Frazer Institute and School of Chemical Engineering, Associate Professor Wolfram will be examining the molecular composition of EVs that are released by triple negative breast cancer cells.
This information will help her team assess how cancer secreted EVs are able to impede natural killer (NK) cells from detecting and destroying cancer cells.

“Armed with this knowledge, we will be in a better place to develop more effective cancer therapies and reduce deaths from this disease,” Associate Professor Wolfram says.
The National Breast Cancer Foundation’s Investigator Initiated Research Scheme offers funding to Australian researchers — both early-career and senior leaders — who have the potential to make significant advances in the field of breast cancer research.
Associate Professor Wolfram has a joint appoitment with the AIBN and UQ's School of Chemical Engineering.
Media: AIBN Communications, Alex Druce, a.druce@uq.edu.au, +61 447 305 979



