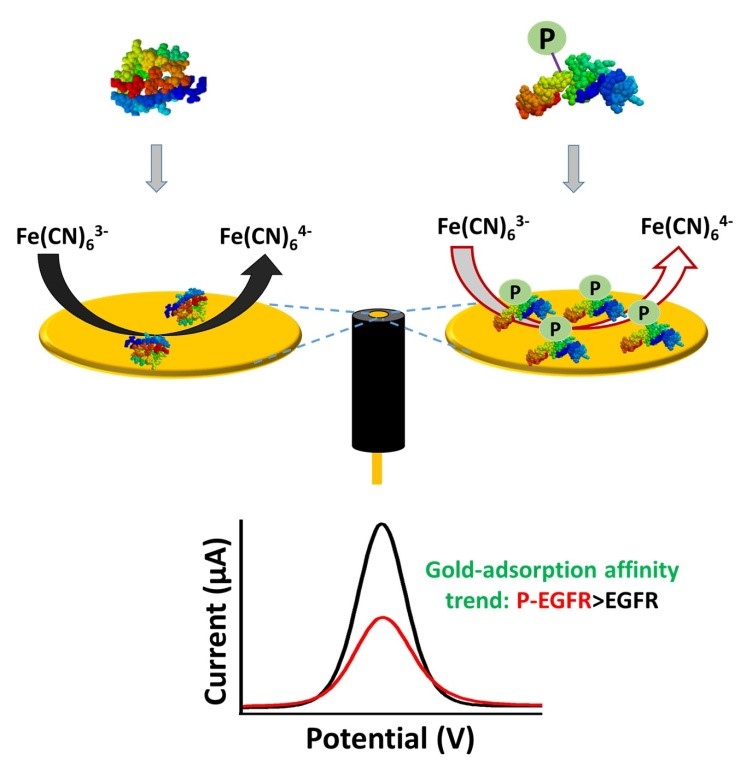
A new interfacial bio-sensing approach for detecting aberrant protein phosphorylation in cancer
Duration:
January 2016–December 2024
Project summary
This project will provide an opportunity for students to acquire skills in molecular biology, biochemistry and electrochemistry.