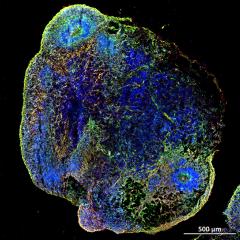
Latest case study
AIBN works with industry to translate cutting-edge science – check out our latest case studies
Meet our industry innovators
AIBN has a unique outward facing operating model that fosters strong industry partnerships to enable translation – watch our videos below to see the impact of our work with our industry partners.
Join The Network
Stay on top of our industry news and developments, events and opportunities, by joining The Network
Precision nanomedicine case studies
AIBN's researchers are examining individual molecules, manufacturing proteins and using the study of bioinformatics to develop treatments getting them from the lab bench to the clinic more quickly than ever.
Nanoengineered materials case studies
AIBN researchers are developing specific solutions to improve existing energy generation technologies, at scale, and develop entirely new ones.
Learn more about nanoengineered materials
Advanced biomanufacturing case studies
AIBN researchers are mapping the processes of biology at the nanoscale, in the gene, genome and protein level of organisms so we can harness cells to create and manufacture new sustainable solutions.
Learn more about advanced biomanufacturing.
Partner with us
Ready to become an industry partner or learn more about what this could mean for you? Get in touch with our dedicated partnerships team today.
Join The Network
Stay on top of our industry news and developments, events and opportunities, by joining The Network